Welcome back to the Smile Science Dental Spa blog! This article will be the first in a series of four articles detailing fluoride, its uses, benefits, risks, and myths. It is based on a narrative review published in Materials earlier this year. Follow along with me, Dr. Richard Dawson, as we explore this topic.
In the grand tableau of elements that grace our planet, fluorine stands out not just for its sheer abundance but also for its distinct characteristics and varied applications. Ranking as the 13th most abundant element in the Earth’s crust, its presence is pervasive. From naturally occurring minerals such as fluorite and cryolite to compounds like fluorapatite and topaz, fluorine’s essence is intricately woven into the planet’s geology.
Beyond its natural occurrence, human activity has further amplified its presence in the atmosphere. Modern industrial advancements, while paving the way for progress, have also introduced fluorides into the environment. Emissions from coal-fired power plants, aluminum works, and even enamel factories contribute to the anthropogenic sources of fluorides. These emissions, in their myriad forms, find their way back to the Earth, settling in our soils or being carried by rain, snow, or fog, potentially infiltrating our ground and surface waters.
Yet, the story of fluoride doesn’t end with its environmental presence. Fluoride is also a crucial micronutrient for mammals. At concentrations of about 500 mg/kg in dry weight, fluoride plays a pivotal role in the structure of supporting tissues, hard tissues of teeth, skin, and hair. Its absorption and metabolism are complex processes, commencing in the oral cavity and continuing through the gastrointestinal tract. Remarkably, within an hour of fluoride consumption, its peak concentration is reached in the bloodstream, and by the subsequent 24 hours, half of it is expelled from the body through various means.
Central to fluoride’s narrative is its undeniable significance in dental health. For decades, dentists worldwide have harnessed the power of fluoride to both prevent and combat dental caries. From toothpaste and floss infused with fluoride to professional dental treatments, fluoride’s role in maintaining oral health cannot be overstated.
Fluoride’s Place in History
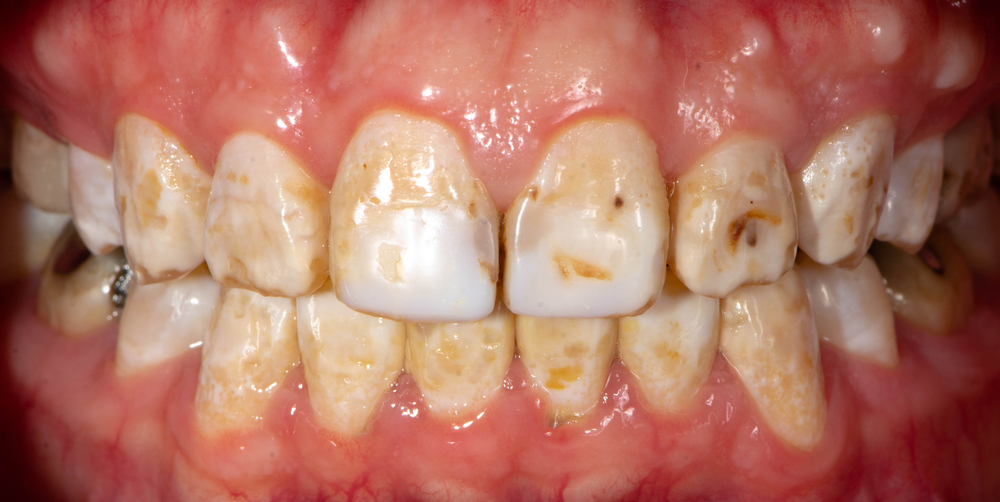
The history of fluoride is as layered and multifaceted as its chemical properties. While its existence and benefits have been known for centuries, its formal recognition and application in the field of dentistry trace back to the 20th century. As stories of certain communities having markedly fewer dental caries began to surface, research pointed to the naturally fluoridated water as the primary reason for such dental resilience.
Initial reactions to the proposed use of fluoride in dentistry were mixed. While the evidence in favor of its caries-preventing properties was compelling, concerns about its safety and potential overuse existed. However, as studies proliferated, showcasing fluoride’s potential in caries prevention, its adoption in dentistry gained momentum.
From its first introduction as an additive in toothpaste to its current widespread use in public water fluoridation projects, fluoride’s journey in dentistry has been transformative. Not only has it reshaped our approach to oral hygiene, but it has also served as a testament to the confluence of tradition, science, and innovation. Its profound impact on dental health, coupled with its rich history, solidifies fluoride’s place as a cornerstone in the annals of dental science.
Cytotoxicity of Fluoride
Fluoride, despite its profound benefits in dental health and its ubiquity in the environment, is not devoid of risks. Its cytotoxic effects, arising from overexposure, can manifest in both acute and chronic scenarios. The boundary that divides beneficial fluoride exposure from harmful is thin, and tipping over into the realm of toxicity can lead to a spectrum of health challenges.
Acute and Chronic Effects of Overexposure
Acute overexposure to fluoride can result in a condition known as fluorosis. Manifested primarily in the dental or skeletal form, fluorosis is indicative of a short-term, high-intensity exposure. Dental fluorosis, often recognized by white streaks or specks on the teeth, reflects excessive fluoride intake during tooth formation. Skeletal fluorosis, on the other hand, is the outcome of longer exposure and affects bones and joints. Chronic exposure to high levels of fluoride can lead to bone fragility, pain, and potentially result in limited joint mobility.
Historical Instances of Fluoride Poisoning and Lessons Learned
Throughout history, there have been incidents that highlighted the risks associated with fluoride. Notable among them was the tragic event in Alaska in 1992 when a water pump malfunctioned, leading to an excessively fluoridated water supply. Many residents suffered symptoms like abdominal pain and vomiting. Such instances were stark reminders of the need for stringent measures to monitor fluoride levels in community water supplies.
From these incidents, valuable lessons were extracted. Monitoring systems were improved, better standards were set, and more extensive education campaigns about the dangers of overexposure were implemented. The emphasis shifted from mere utilization to responsible and informed use.
Acute Poisoning with Fluoride Compounds

Fluoride compounds are found in various settings, from household products to industrial applications. While the benefits of fluoride are many, so too are the risks when it comes into the equation in disproportionate amounts.
Causes and Examples of Acute Poisoning:
Acute poisoning with fluoride compounds can result from ingestion, inhalation, or dermal exposure to significant amounts of these compounds. Common sources include dental products, insecticides, rodenticides, and certain industrial chemicals. Accidental ingestion of fluoride tablets, excessive consumption of fluoridated water, or inhalation of fluoride fumes from industrial processes are some examples.
Toxic Dose Thresholds and Effects on the Human Body:
The toxic threshold for fluoride varies based on the route of exposure and individual differences. However, acute symptoms often manifest after ingestion of doses exceeding 5 mg/kg of body weight. Initial symptoms can include nausea, vomiting, diarrhea, and abdominal pain. In more severe cases, individuals may experience muscle spasms, difficulty in breathing, cardiac abnormalities, and even coma or death.
Long-term exposure to high levels of fluoride can have a range of effects on the human body, from the aforementioned fluorosis to potential neurological impacts. While the body possesses mechanisms to excrete excess fluoride, primarily via urine, persistent exposure can overwhelm these systems, leading to accumulation and resultant health challenges.
Awareness, education, and stringent controls are essential to minimize the risks associated with fluoride compounds. As with many substances, it’s a matter of balance—leveraging the benefits while staying vigilant about the potential dangers.
Understanding Fluoride Dosage and Toxicity

Fluoride, an element essential for dental health when consumed in the right amounts, can become a potential health hazard when the balance tilts towards excess. Thus, understanding the appropriate dosage, especially for varying age groups, is crucial.
Safe Levels for Different Age Groups:
The optimal fluoride levels depend on multiple factors, such as age, dietary habits, body weight, and overall health. Here are general guidelines, but it’s important to note that recommendations might vary based on regional factors, such as the fluoride concentration in local water sources:
- Infants (0-6 months): No supplemental fluoride is necessary, as they primarily consume breast milk or formula.
- Babies (6-12 months): Up to 0.01 mg/day, especially if they consume non-fluoridated water.
- Children (1-3 years): 0.7 to 1.2 mg/day.
- Children (4-8 years): 1 to 2.2 mg/day.
- Children (9-13 years): 1.5 to 2.5 mg/day.
- Teenagers (14-18 years): 1.3 to 2.7 mg/day for females and 1.5 to 3.1 mg/day for males.
- Adults: 1.5 to 4 mg/day, with the upper limit being especially for those living in non-fluoridated areas.
The Relationship Between Dose and Toxicity:
The body can tolerate certain amounts of fluoride without any adverse effects. However, as the dosage increases, the risk of toxicity also rises. Acute fluoride toxicity can manifest with doses as low as 5 mg/kg of body weight. Chronic overexposure can lead to dental and skeletal fluorosis, among other potential health complications. It’s a classic case of “the dose makes the poison”; what is beneficial in controlled amounts can become harmful when consumed in excess.
Fluoride in Dental Prophylaxis
Fluoride’s role in dental prophylaxis, especially in preventing dental caries, has been extensively documented and is the primary reason for its incorporation into dental care products.
The Role of Fluoride in Preventing Dental Caries:
Fluoride aids in the remineralization of tooth enamel, making it more resistant to acid attacks from bacteria in the mouth, thereby preventing the onset and progression of cavities. When fluoride is present, it gets incorporated into the tooth structure, resulting in fluorapatite, which is more acid-resistant than the original tooth mineral, hydroxyapatite.
Different Fluoride-based Products and Their Applications
- Toothpaste: The most common source of fluoride for many individuals. Regular use helps maintain a constant level of fluoride in the mouth.
- Mouth Rinses: Over-the-counter fluoride mouthwashes can be used for additional protection, especially for those at a higher risk of dental caries.
- Gels and Foams: Applied by dental professionals, these offer a higher fluoride concentration, providing a protective layer to the teeth.
- Varnishes: Professionally applied, fluoride varnishes offer prolonged fluoride release, helping in the prevention of dental decay.
- Supplements: In areas with low natural fluoride levels in the drinking water, fluoride supplements (tablets, drops) may be prescribed, especially for children.
It’s essential to remember that while fluoride plays a pivotal role in dental health, moderation is key. With appropriate dosing and application, the benefits of fluoride can be reaped without veering into the territory of toxicity.
Natural Sources of Fluoride

Fluoride’s presence in nature is vast, with it originating from the Earth’s crust and finding its way into various natural systems, including water, soil, and some food items.
Geological Sources, Water Sources, and Food Items Rich in Fluoride:
- Geological Sources: Fluorite (calcium fluoride) is the primary mineral source of fluoride. Other minerals containing fluoride include cryolite, apatite, and mica.
- Water Sources: Fluoride can be found naturally in fresh and saltwater sources. Groundwater, particularly from deep wells, often contains higher fluoride levels than surface water.
- Food Items: Certain foods naturally contain fluoride. Tea leaves, for instance, accumulate fluoride from soil and water. Fish, especially those with edible bones like sardines, and certain marine food products also have high fluoride concentrations.
Regions with Naturally High Fluoride Concentrations:
Certain regions worldwide, including parts of India, China, East Africa, and the Rift Valley countries, have been identified with high fluoride concentrations in their groundwater. These high levels can sometimes result in endemic fluorosis among local populations.
Industrial and Man-made Sources of Fluoride

In the age of industrialization, numerous processes have introduced additional fluoride into the environment.
Fluoride in Industry and its Impact on the Environment
- Aluminum Production: The production of aluminum from bauxite ore releases gaseous fluoride compounds.
- Phosphate Fertilizer Production: The production of phosphate fertilizers can release hydrogen fluoride and silicon tetrafluoride.
- Steel and Iron Industries: Certain processes in these industries release fluoride-containing gases and particulates.
- Coal Combustion: Burning coal releases fluoride, which can be deposited on land and absorbed by plants or enter water systems.
- Oil Refineries: These can release gaseous fluorides as part of their refining processes.
- Fluorinated Hydrocarbon Production: Industries producing refrigerants, propellants, and solvents may release various fluoride compounds.
Controlling Emissions and Managing Fluoride Pollution
- Scrubbers: Installing wet or dry scrubbers in industries helps capture gaseous fluoride compounds before they’re released into the atmosphere.
- Settling Chambers: These allow larger fluoride particulates to settle, preventing their release.
- Lime Addition to Water: By adding lime to water sources, soluble fluoride is converted into insoluble calcium fluoride, which can be removed.
- Regulations and Guidelines: Government and environmental bodies worldwide have established guidelines for permissible fluoride emissions. Regular monitoring and adherence to these standards can significantly reduce environmental fluoride pollution.
- Public Awareness: Educating the public about the sources and potential dangers of fluoride pollution encourages better practices and urges industries to adopt eco-friendly methods.
Ensuring the balance between utilizing fluoride’s benefits and minimizing its potential environmental impact requires a concerted effort between industries, governments, and the general public.
Managing and Mitigating Fluoride Exposure:

- Monitor Drinking Water: Regularly test the fluoride levels in drinking water, especially in regions known for high natural fluoride content. Utilize water filters that effectively reduce fluoride if levels are excessively high.
- Limit Consumption of Fluoride-rich Foods: While some fluoride intake is beneficial, excessively consuming fluoride-rich foods like tea and certain fish can be harmful. It’s essential to have a balanced diet.
- Use Fluoridated Dental Products Judiciously: Especially in areas with fluoridated water, monitor the use of fluoride toothpaste and mouth rinses, especially among children.
- Be Aware of Workplace Exposure: If you work in industries that deal with fluoride, take precautions to limit exposure. Use protective gear and ensure good ventilation in work areas.
Treatment and Preventive Measures for Fluoride Poisoning
- Immediate Intervention: In case of acute fluoride ingestion, induce vomiting and seek emergency medical attention immediately.
- Calcium Therapy: Administering calcium-based compounds, like calcium gluconate, can help counteract fluoride toxicity by binding to fluoride.
- Dialysis: In severe cases, hemodialysis might be required to remove excess fluoride from the bloodstream.
- Regular Health Check-ups: People living in areas with high fluoride concentrations should get regular health check-ups, especially focusing on bone health and dental condition.
- Educational Programs: Implement community-based programs to educate people about the sources, risks, and management of excessive fluoride exposure.
The Future of Fluoride in Dentistry
- Nano-fluoride Applications: Recent research suggests that nanoparticle-sized fluoride can penetrate deeper into tooth enamel, providing more profound and lasting protection against cavities.
- Smart Toothbrushes: Technological advances are leading to toothbrushes that can dispense the right amount of fluoride, ensuring optimal protection without overexposure.
- Tailored Fluoride Treatments: Using genetic and personalized data to determine the exact fluoride requirements for individuals can lead to more effective and safer dental treatments.
The Balance Between Health Benefits and Potential Risks: Fluoride’s role in dentistry has undeniably revolutionized dental health, reducing the prevalence of cavities and improving overall oral hygiene. However, the delicate balance between its benefits and potential adverse effects has always been a concern. As science and technology advance, dentistry must pivot towards methods that optimize fluoride’s positive aspects while mitigating its risks. This requires continuous research, public awareness campaigns, and adopting a holistic approach that considers individual needs and environmental impacts.
Further Reading
This article is the first of four articles in our Fluoride Series. It was adapted and written from the following article, published in Materials in February, 2023:
The Safety of Fluoride Compounds and Their Effect on the Human Body—A Narrative Review