The significance of fluoride in the context of human health and development has evolved over centuries. From the early days of its discovery to its vast applications today, fluoride remains one of the most debated and researched elements.
Historical Significance of Fluoride
Fluoride is not a new entrant in the world of science. Historically, its presence was noted in natural water sources, and certain communities consuming this water demonstrated remarkably lower rates of dental caries. It wasn’t until the early 20th century, however, that the scientific community began rigorously investigating this phenomenon. They found that the levels of fluoride in water had a direct correlation to the dental health of the population consuming it.
Initial Recognition in the Field of Dentistry
The early 1900s marked a pivotal era for dentistry. Upon observation of the aforementioned phenomenon, researchers hypothesized that fluoride could be beneficial for dental health. This hypothesis was met with a series of studies aiming to ascertain the role of fluoride in preventing tooth decay. It didn’t take long before dentists across the globe recognized the potential of fluoride. Its ability to strengthen tooth enamel, prevent cavities, and even reverse early stages of tooth decay propelled fluoride to the forefront of preventive dentistry. Today, it’s hard to imagine a dental care regimen without some reference to fluoride, a testament to its immense importance in the field.
Sources of Fluoride

Fluoride, as an element, can be found in numerous sources. Its presence can be attributed to both natural processes and human-made interventions. An understanding of these sources is pivotal in grasping the extensive reach and influence of fluoride in our environment and daily lives.
Natural Sources
Water: Fluoride naturally occurs in water, with concentrations varying based on regional geological formations. Some areas are fortunate to have the optimal level of fluoride, beneficial for dental health, while others might have levels either too low or excessively high.
Food: Several foods, especially those grown in fluoride-rich soils, contain this mineral. Tea, for instance, has been known to have a considerable amount of fluoride, especially if grown in certain regions.
Air: Trace amounts of fluoride can be found in the air, usually originating from soil and rocks.
Anthropogenic (Human-made) Sources
Industrial: Many industries, including aluminum and steel production, coal-burning, and phosphate fertilizer manufacturing, release fluoride as a byproduct. This can lead to elevated levels of fluoride in nearby water sources and the surrounding environment.
Agricultural: The use of pesticides and fertilizers can sometimes introduce fluoride into the environment. Certain pesticides contain fluoride compounds to enhance their effectiveness, and these can leach into the soil and water, impacting the fluoride levels in crops and potentially entering the food chain.
Mechanisms of Fluoride Action
Fluoride’s remarkable ability to protect and enhance dental health is not a mere coincidence. There is a precise and scientific basis for its effectiveness. Here are some of the key mechanisms through which fluoride acts:
Formation of Fluorapatite
Fluorapatite is a naturally occurring mineral in teeth. When fluoride is present, it reacts with the hydroxyapatite crystals in tooth enamel to form fluorapatite. This compound is more resistant to acid attacks, which means teeth are better protected against decay-causing bacteria. The transformation ensures that the enamel becomes harder and less soluble in acidic environments.
Inhibition of Demineralization
Tooth decay begins when acid-producing bacteria in the mouth attack the enamel, leading to demineralization. Fluoride, when present, helps reduce the rate at which this enamel dissolves in response to these acid attacks, ensuring that the tooth remains stronger for longer.
Enhancement of Remineralization
Fluoride doesn’t just inhibit the loss of minerals; it also promotes the addition of minerals back into the tooth enamel. This process, called remineralization, helps in repairing early stages of tooth decay and can reverse minor tooth damages before they become cavities.
Bacterial Enzyme Inhibition
Fluoride’s action isn’t limited to tooth structures. It also impacts bacteria directly. Some strains of bacteria in the mouth produce enzymes essential for their metabolism. Fluoride can inhibit these enzymes, limiting the bacteria’s ability to produce acids, hence reducing the potential for tooth decay.
Modes of Fluoride Application
To optimize fluoride’s benefits, various methods have been developed to ensure its delivery to the population. These can be broadly categorized into endogenous and exogenous methods:
Endogenous Fluoridation
Through Drinking Water: One of the most widespread methods, water fluoridation involves adjusting fluoride levels in community water supplies. This provides a consistent and passive means for people to receive fluoride’s benefits.
Through Milk and Salt: Certain regions introduce fluoride to milk or salt as a public health strategy. Like water fluoridation, it ensures regular intake and provides benefits especially in areas where water fluoridation is not feasible.
Exogenous Fluoridation
Direct Action on Tooth Surface: Direct application methods focus on putting fluoride compounds in direct contact with teeth. This can be achieved through various means:
- Brushing: Most toothpastes contain fluoride, and brushing ensures direct contact with teeth.
- Rinsing: Mouthwashes with fluoride offer additional protection, especially in areas with low fluoride levels in water.
- Flossing: Some dental flosses are also infused with fluoride, offering protection between teeth.
- Compressing: Fluoride gels or varnishes can be applied using trays or directly onto the teeth, usually in dental offices.
Mention of Various Compounds (Organic and Inorganic)
Fluoride can be found in both organic and inorganic compounds. The most commonly used inorganic fluoride compounds include sodium fluoride (NaF), stannous fluoride (SnF2), and acidulated phosphate fluoride (APF). Organic fluoride compounds, on the other hand, include monofluorophosphate, often found in toothpaste.
Professional Procedures
Professional dental offices offer an array of fluoride treatments that are higher in concentration compared to over-the-counter products. These treatments provide an intensive boost to dental health, especially for individuals at high risk for cavities.
High-concentration Fluoride Treatments:
- Fluoride Varnishes: Often containing 5% sodium fluoride, these varnishes are applied directly to the teeth. They adhere to the teeth’s surface, releasing fluoride over time.
- Fluoride Gels: Typically available in 1.23% acidulated phosphate fluoride (APF) or 2% sodium fluoride varieties, these gels can be applied using trays or brushed directly onto teeth.
- Fluoride Foams: Similar in concentration to gels, foams are applied using trays, allowing for efficient fluoride release to all tooth surfaces.
Home Treatments
While professional treatments offer high fluoride concentrations, daily oral care routines also play a pivotal role in maintaining dental health. This is where home treatments come into play:
- Toothpastes: Most toothpastes contain fluoride, usually in the form of sodium fluoride or monofluorophosphate. Their concentrations typically range between 1000 to 1450 ppm.
- Rinses: Mouthwashes and rinses with fluoride offer supplementary protection. They are particularly beneficial for those who live in areas with non-fluoridated water or for individuals prone to cavities. Over-the-counter fluoride rinses usually contain lower concentrations than prescription versions.
Potential Hazards
Fluoride, while beneficial in the right amounts, can pose risks when consumed excessively:
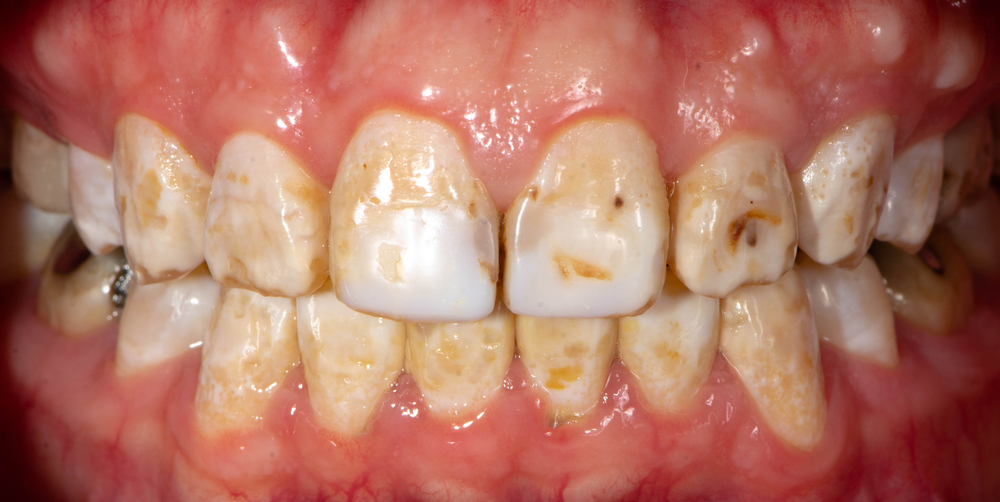
- Fluoride Accumulation: Chronic ingestion of excessive fluoride can lead to a condition called dental fluorosis. While usually cosmetic and characterized by white spots on teeth, severe fluorosis can cause enamel pitting or discoloration.
- Acute Poisoning Risks: Ingesting large amounts of fluoride in a short period can lead to fluoride poisoning. Symptoms may include nausea, vomiting, diarrhea, abdominal pain, and increased salivation. In extreme cases, it can lead to muscle spasms, cardiac arrest, and even death. It’s essential for fluoride-containing products to be stored out of reach of children to prevent accidental ingestion.
Fluoride Mouth Rinse

Fluoride mouth rinses are specially formulated solutions that contain fluoride to prevent tooth decay and strengthen tooth enamel.
Benefits of Fluoride-Enhanced Rinses
- Tooth Decay Prevention: Regular use can help reduce the onset of cavities.
- Enamel Strengthening: Fluoride in the rinse promotes remineralization of the enamel, helping counteract the effects of acidic foods and drinks.
- Sensitive Teeth Relief: Some fluoride mouth rinses can help reduce tooth sensitivity by fortifying exposed root surfaces.
Recommendations for Usage
- Frequency: Daily or weekly use, based on fluoride concentration and individual dental needs.
- Post-Rinse Protocol: Avoid eating or drinking for 30 minutes after rinsing to maximize fluoride contact with teeth.
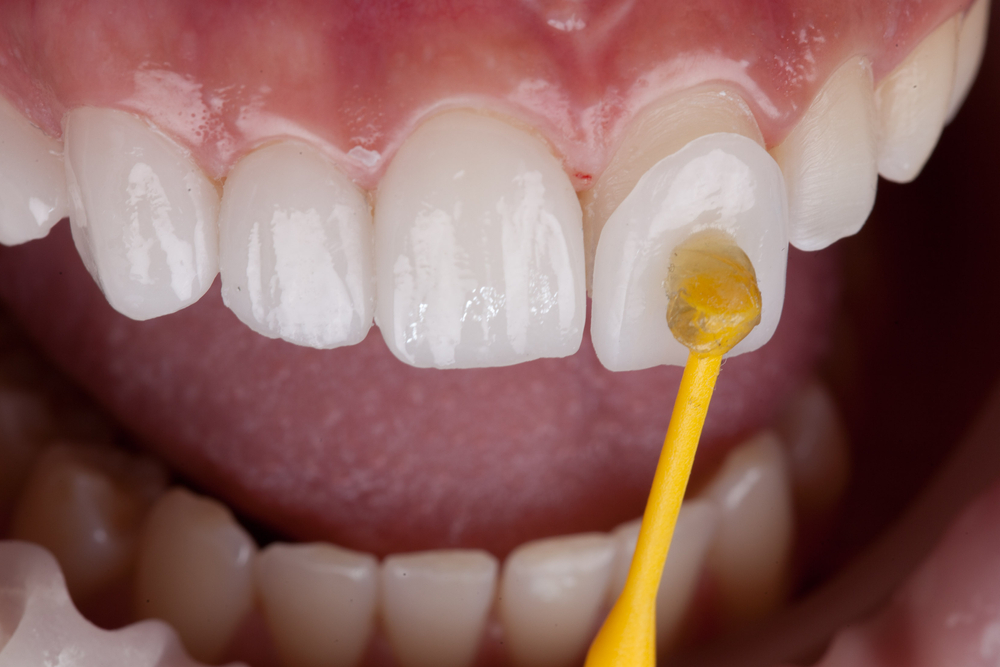
Fluoride Varnish
Fluoride varnishes have gained popularity in dental offices as an effective topical fluoride treatment.
Composition and Mechanism
- Composition: Typically comprised of 5% sodium fluoride in a resin base.
- Mechanism: The varnish adheres to tooth surfaces, slowly releasing fluoride over an extended period, allowing for prolonged tooth enamel interaction.
Application and Benefits
- Application: Applied directly to teeth using a brush; sets rapidly upon contact with saliva.
- Benefits:
- Prolonged fluoride release ensures extended protection.
- Effective for individuals with high cavity risk.
- Can help reduce tooth sensitivity.
Fluoride Foam and Gel
Both fluoride foam and gel are used in professional settings for fluoride application, but they differ in their consistency and sometimes in their method of application.
Comparison in Fluoride Concentration
- Foam: Typically has a concentration similar to gels, often around 1.23% acidulated phosphate fluoride (APF) or 2% sodium fluoride.
- Gel: Available in concentrations such as 1.23% APF or 2% sodium fluoride.
Application Methods
- Foam: Applied using a tray that fits around the teeth, allowing the foam to reach all tooth surfaces.
- Gel: Can be applied with trays like foam or brushed directly onto the teeth.
Comparison in Efficacy with Varnishes
- While both foams and gels offer significant fluoride exposure, varnishes generally provide longer-lasting fluoride contact with tooth surfaces due to their adhesive nature. This prolonged contact can lead to more effective prevention of decay and enamel strengthening.
Filling Materials
Dental fillings are essential in restorative dentistry, not only for restoring the function and structure of a tooth but also for delivering therapeutic agents like fluoride.
Types of Fluoride-Releasing Materials
- Glass Ionomer Cements (GIC): These are tooth-colored materials that release fluoride over time, helping prevent decay around the filling.
- Resin-modified Glass Ionomer Cements: A hybrid of the traditional GIC with added resin for improved strength and aesthetics.
- Compomers: Materials that combine the properties of composite resins and glass ionomers. They release fluoride and have the added advantage of being tooth-colored.
- Fluoride-releasing Amalgams: Traditional amalgams infused with fluoride.
Benefits in Restorative Dentistry
- Long-Term Fluoride Release: Continuous release of fluoride aids in preventing secondary caries around the restorations.
- Biocompatibility: Fluoride-releasing materials tend to be biocompatible and promote tooth health.
- Adhesion: Materials like GICs bond well with tooth structures, providing longevity to the restoration.
Fissure Sealants
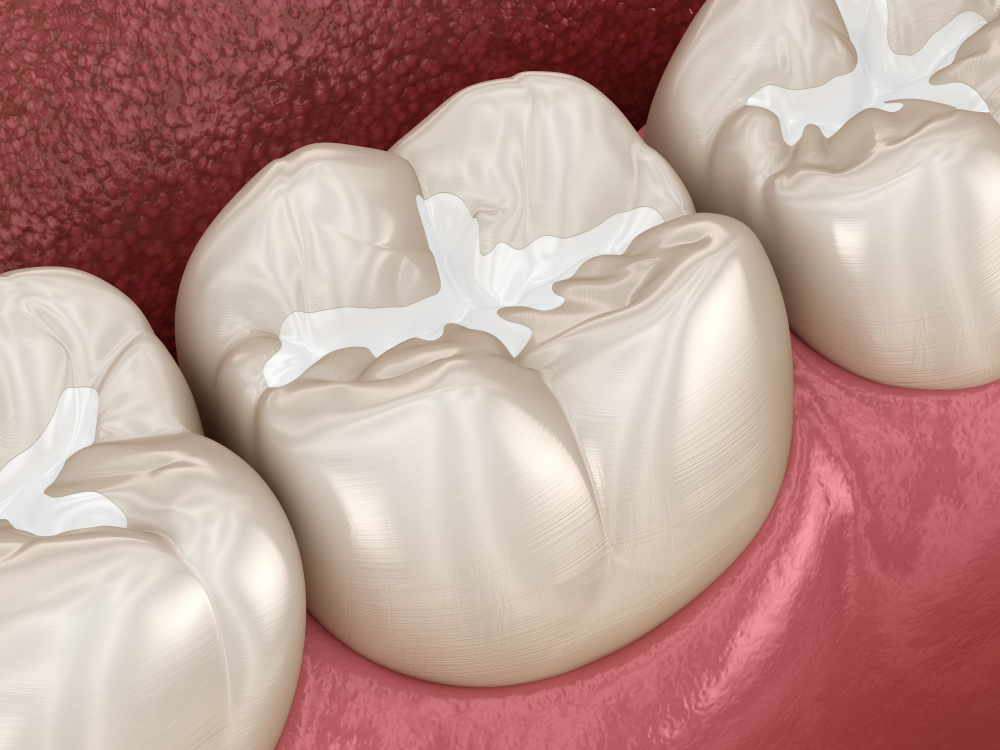
Fissure sealants are protective coatings applied to the occlusal (biting) surfaces of teeth to prevent decay.
Rationale for Use
- Protection of Deep Grooves: Molars and premolars often have deep grooves that can trap food and bacteria, making them susceptible to cavities.
- Childhood Caries Prevention: Sealants are particularly effective for children who are at high risk of cavities.
Benefits in Caries Prevention
- Physical Barrier: Sealants act as a barrier, keeping bacteria and food particles away from tooth surfaces.
- Long-Lasting Protection: Once applied, sealants can protect teeth for many years.
- Fluoride Release: Some sealants are fluoride-infused, further boosting their protective qualities.
Medicines and Agents Used in Medicine
Beyond dentistry, fluoride has medical applications, particularly in bone health and diagnostic imaging.
Fluoride’s Role in Osteoporosis Treatment
- Bone Density Enhancement: Fluoride has been explored for its potential to increase bone density, making it a candidate for osteoporosis management.
- Stimulating Bone Formation: Fluoride can stimulate the growth of osteoblasts, the cells responsible for bone formation.
Diagnostic Applications
- PET Scans: Fluorine-18, a radioactive isotope of fluorine, is used as a tracer in Positron Emission Tomography (PET) scans, which help visualize internal body processes including metabolism and blood flow.
Fluoride’s diverse roles in both dentistry and medicine highlight its importance and versatility in promoting health and aiding in diagnosis.
Industrial and Economic Development
As industries evolve and economies grow, so do their environmental footprints. One significant aspect of this is the release of various chemicals, including fluoride, into the environment.
Environmental Implications of Fluoride Release:
- Water Contamination: Direct discharges from industries can lead to high concentrations of fluoride in surface and groundwater, affecting both human populations and aquatic life.
- Air Pollution: Fluoride emissions from certain industries can contaminate the air, leading to inhalation risks for populations residing nearby.
Major Sources of Fluoride Emissions
- Aluminum Production: This industry is a primary source of fluoride emissions due to the smelting process.
- Phosphate Fertilizer Production: The processing of phosphate rock releases significant amounts of fluoride.
- Coal Burning: When coal is burnt, it releases fluoride into the atmosphere.
Carcinogenic Risks Associated with Benzofluorants
- While fluoride itself is not considered carcinogenic, some fluoride-containing compounds, like certain benzofluorants, have been under scrutiny for potential cancer risks.
Regional Implications
- Fluorosis in China: In some regions of China, high levels of naturally occurring fluoride in drinking water have led to widespread dental and skeletal fluorosis among residents.
Agriculture

The interplay between agriculture and fluoride is intricate, with fluoride being both a component of certain agricultural products and affected by them.
Fluoride Content in Plants
- Natural Accumulation: Some plants can accumulate fluoride from the soil, especially if the soil has high fluoride content.
- Bioaccumulation: Prolonged exposure to fluoride-contaminated water or soil can result in plants storing significant amounts of fluoride.
Impacts of Pesticides and Fertilizers on Fluoride Levels
- Pesticides: Some pesticides may contain fluoride or increase the plant’s uptake of fluoride from the environment.
- Fertilizers: Phosphate fertilizers, in particular, can contribute to elevated fluoride levels in soil, subsequently affecting crops.
Conclusion
Fluoride, a naturally occurring element, has embedded itself deeply into modern society. Its roles range from enhancing dental health to its presence in industrial emissions and agricultural products.
Overall Significance of Fluoride in Modern Society
- Health Benefits: The primary benefit of fluoride remains its ability to enhance dental health and potentially treat conditions like osteoporosis.
- Industrial Importance: Fluoride’s role in industries such as aluminum production is significant.
Balancing Benefits with Potential Risks
- While the health benefits of fluoride, particularly in dental care, are well-established, there are genuine concerns regarding overexposure and associated health implications like fluorosis.
Future Prospects and Areas for Research
- The continued research into fluoride’s potential risks and benefits, its environmental impacts, and its broader roles in industries and agriculture is crucial for informed decision-making and ensuring the element is used safely and effectively.
Further Reading
This article is the third of four articles in our Fluoride Series. It was adapted and written from the following article, published in Materials in February, 2023:
The Safety of Fluoride Compounds and Their Effect on the Human Body—A Narrative Review